Why Pharmaceutical Cold Storage is Critical for Patient Safety
Pharmaceutical cold storage is a specialized system that maintains precise temperature conditions to preserve the safety, efficacy, and integrity of temperature-sensitive medications, vaccines, and biological products. Here's what you need to know:
Key Requirements:
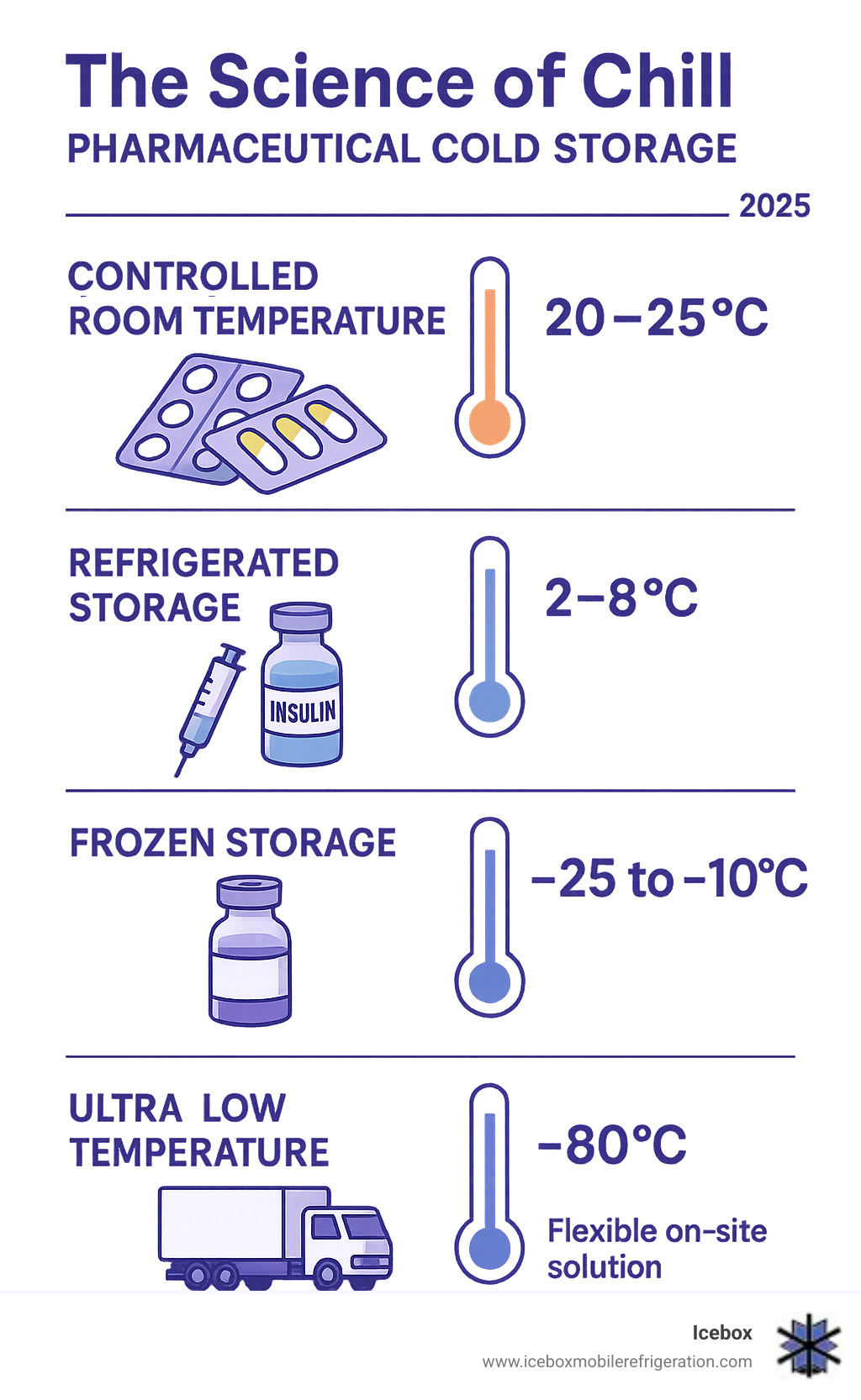
- Temperature ranges: 2°C to 8°C for most vaccines and biologics, -25°C to -10°C for frozen products
- Continuous monitoring: 24/7 temperature tracking with real-time alerts
- Regulatory compliance: Must meet FDA and Good Distribution Practices (GDP) standards
- Product protection: Prevents degradation of biologics, vaccines, insulin, and cancer treatments
The stakes couldn't be higher. When pharmaceutical products are exposed to temperature excursions, they can lose potency or become dangerous to patients. A significant percentage of temperature-sensitive medicines are reportedly impacted by temperature excursions, leading to millions in losses and potential harm to patients.
The pharmaceutical industry has become increasingly dependent on cold storage. Medicines requiring cold storage now represent 35% of the pharmaceutical market - up from just 26% in 2017. With approximately half of all new medicines expected to require cold storage in the next five years, the demand for reliable, flexible cold storage solutions continues to grow.
Modern healthcare facilities face unique challenges: power outages, equipment failures, seasonal demand spikes, and facility renovations. These situations require on-site mobile cold storage solutions that can be deployed quickly to maintain the cold chain without interruption.
Why Precise Temperature Control is Non-Negotiable for Pharmaceuticals
The world of pharmaceuticals is a delicate ecosystem where every degree truly matters. The integrity of medications, vaccines, and other biological products hinges entirely on maintaining specific, often narrow, temperature ranges. This isn't just a best practice; it's a fundamental requirement for drug stability, preventing contamination, extending shelf life, and ensuring vaccine efficacy. It’s about avoiding financial loss and, most importantly, safeguarding public health.
Many pharmaceutical products, including vaccines, antibiotics, and biologics, are incredibly sensitive to temperature fluctuations. These products, whether finished medicines, specialized therapies like gene therapy, or even active pharmaceutical ingredients (APIs), can lose their effectiveness and even become unsafe if exposed to extreme temperatures. Think of it like a carefully crafted recipe: if you mess with the ingredients or the cooking temperature, the final dish just won't be right.
Preserving Drug Efficacy and Safety
At the heart of pharmaceutical cold storage is the imperative to preserve drug efficacy and safety. Without precise temperature control, sensitive compounds can undergo chemical degradation or protein denaturation. This means that the active ingredients, which are responsible for the drug's therapeutic effects, can break down or change their structure. When this happens, the medication loses its intended potency, rendering it less effective or completely ineffective.
Consider modern biologic therapies, such as insulin, monoclonal antibodies, and advanced cellular and gene therapies. These are often complex proteins or living cells that are highly susceptible to temperature changes. If not stored correctly, they can denature, or essentially "unfold," losing their activity and becoming useless. This poses a significant patient risk, as a compromised drug means a compromised treatment, potentially leading to adverse health outcomes or a complete failure of therapy. Improper storage can even lead to the formation of harmful byproducts, turning a life-saving medication into a dangerous one.
Meeting Strict Regulatory and Financial Imperatives
Beyond the immediate impact on patient safety, the pharmaceutical industry operates under a stringent web of regulatory and financial imperatives. Regulatory authorities, such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), mandate appropriate temperature conditions for pharmaceutical storage. Good Distribution Practices (GDP) and current Good Manufacturing Practices (cGMP) are not just suggestions; they are legally binding frameworks that ensure product quality and integrity.
Failure to comply with these standards can have catastrophic consequences. Improper storage can lead to massive inventory loss from wasted product or harm to patients from out-of-date or improperly stored drugs. This isn't just a theoretical concern: a significant percentage of temperature-sensitive medicines are reportedly impacted by temperature excursions, leading to significant financial losses. The global expenditure on biopharma cold storage is projected to reach an astounding 21.3 billion USD this year, a substantial increase from 15.4 billion USD in 2018. This growing investment underscores the critical financial stakes involved.
A single temperature deviation can compromise an entire batch of medication, resulting in costly recalls, irreparable reputational damage, and, in severe cases, legal ramifications. The ability to maintain precise temperature control is not just good practice; it's essential for market access, avoiding regulatory penalties, and preserving financial stability. For more detailed information on maintaining temperature control, you can explore our resources on Refrigerated Storage. For helpful hints and practical advice, check out our Cold Storage Tips.
Navigating Key Temperature and Regulatory Standards
The world of pharmaceutical cold storage is defined by specific temperature zones, each custom to the unique stability requirements of different medications. Understanding these zones and the regulatory standards that govern them is paramount for ensuring product integrity.
Standard Pharmaceutical Cold Storage Temperatures
Regulatory bodies like the United States Pharmacopeia (USP) provide precise definitions for medicine storage and shipment temperatures. These guidelines dictate everything from ambient conditions to ultra-low freezing, ensuring that products remain stable from manufacturing to patient administration.
Here's a breakdown of the standard temperature ranges and the types of products typically stored within them:
| Temperature Range (Celsius/Fahrenheit) | USP Definition | Typical Products Stored |
|---|---|---|
| 20°C–25°C (68°–77°F) | Controlled Room Temperature | Tablets, capsules, some solutions, non-refrigerated biologics |
| 8°C–15°C (46°–59°F) | Cool | Certain oral medications, some medical devices |
| Not over 8°C (Not over 46°F) | Cold | General reference for cold conditions |
| 2°C–8°C (36°–46°F) | Refrigerator | Most vaccines, insulin, biologics (e.g., monoclonal antibodies), some antibiotics, clinical trial materials |
| -10°C to -25°C (14°F to -13°F) | Freezer | Certain vaccines, some biologics, specific diagnostic reagents |
| Below -25°C (Below -13°F) | Deep Freezer / Ultra-low | mRNA vaccines (e.g., -70°C), cellular and gene therapies, some APIs, critical research samples |
| Below -150°C (Below -238°F) | Cryogenic | Cell lines, tissues, highly sensitive biological samples (often in liquid nitrogen vapor phase) |
The most common requirement for temperature-sensitive pharmaceuticals is the "refrigerated" range of 2°C to 8°C. This is the sweet spot for many vaccines, biologics, and insulin, where stability is maximized without freezing. However, the rise of advanced therapies has driven a significant increase in demand for ultra-low temperature storage, sometimes as low as -70°C or even -196°C, as seen with some mRNA vaccines and cellular therapies.
The Role of Validation and Monitoring
It's not enough to simply have a cold space; you need to prove it works consistently and reliably. This is where validation and continuous monitoring come into play.
- Temperature Mapping: Before a cold storage unit is used for pharmaceuticals, it undergoes a rigorous process called temperature mapping. This involves placing numerous sensors throughout the unit to identify any hot or cold spots and ensure uniform temperature distribution.
- Data Loggers and Alarm Systems: Once validated, cold storage units are equipped with sophisticated data loggers that continuously record temperature data. These systems are often connected to real-time alarm systems that trigger alerts via email, text, or even phone calls if the temperature deviates from the set range. This immediate notification allows for rapid intervention, preventing potential product loss.
- Continuous Monitoring: The best practice in pharmaceutical cold storage is 24/7 continuous monitoring. This provides an unbroken audit trail of temperature conditions, which is crucial for regulatory compliance and product traceability.
- Calibration: All monitoring equipment, including sensors and data loggers, must be regularly calibrated against national standards to ensure accuracy. This ensures that the recorded data is reliable and trustworthy.
- Compliance Documentation: Every step of the validation and monitoring process must be carefully documented. This comprehensive documentation is essential for regulatory audits and demonstrating adherence to GDP guidelines.
For more information on the precise definitions and standards, you can refer to the Official USP temperature definitions. To understand how this applies to our temperature-controlled solutions, take a look at our insights on Climate Controlled Storage Rental.
Overcoming Problems in the Pharmaceutical Cold Chain
The journey of a pharmaceutical product, from its creation to reaching a patient, is a careful and complex one. Even with the best plans, challenges often pop up, especially when it comes to on-site storage – that crucial point when medications are waiting to be used at hospitals, pharmacies, or clinics. Maintaining pharmaceutical cold storage at every step is absolutely vital, but real-world situations can throw a wrench in the works.
The Challenge of On-Site and Emergency Storage Needs
Even the most carefully designed cold chain can face unexpected bumps in the road. These often lead to urgent needs for on-site and emergency cold storage that traditional solutions simply can't handle fast enough. Imagine these tricky situations:
- Power Outages: A sudden loss of electricity can quickly turn precious, temperature-sensitive medications into unusable waste. Without a quick backup, valuable pharmaceuticals can be ruined in just a few hours.
- HVAC Failure: If your facility's heating, ventilation, and air conditioning system breaks down, it can put your entire inventory at risk, not just in your main storage but even in your controlled room temperature areas.
- Inventory Overflow: Sometimes, you just need more space! Whether it's during a busy season like flu vaccine distribution or an unexpected surge in product deliveries, your existing cold storage might not be enough. This often leads to a frantic search for extra space that meets strict temperature rules.
- Renovations: When a hospital, pharmacy, or lab is undergoing construction or upgrades, their regular cold storage units might need to be moved or shut down temporarily. This creates an immediate need for a compliant, temporary solution.
- Special Projects: Think about large-scale immunization campaigns, clinical trials, or the temporary holding of specialized therapies. These projects often require dedicated, temporary cold storage that can be brought right to where it's needed, exactly when it's needed.
- Public Health Crises: As we've all seen, global health events can create huge, urgent demands for specialized cold storage, sometimes even ultra-low temperatures, and rapid distribution.
In these crucial moments, being able to quickly bring in reliable, on-site cold storage isn't just convenient; it's absolutely essential for keeping the cold chain intact and protecting public health. Failing to do so can result in huge financial losses and, more importantly, put patients at serious risk. For more on how we can help in these situations, check out our Cold Storage Delivery Service and learn about our Emergency Cold Storage options.
Flexible Solutions for Modern Pharmaceutical Cold Storage
This is precisely where Icebox Mobile Refrigeration steps in! We know that a one-size-fits-all approach just doesn't cut it in the world of pharmaceuticals. Our mobile refrigerated trailers and mobile freezer trailers are designed to tackle these challenges head-on, giving you the flexibility, reliability, and peace of mind you need.
Our solutions offer a practical way to manage overflow, emergencies, and special projects. We provide on-site placement, meaning we deliver our mobile cooler trailers right to your hospital, pharmacy, clinic, or research facility. This keeps your critical pharmaceuticals safely on your premises, easily accessible, and under your direct control. You avoid the risks that come with moving products to an off-site facility. Our solutions also offer incredible scalability; whether you need a small unit for temporary overflow during flu season or several trailers for a major renovation, we can meet your exact capacity needs. When an emergency hits, rapid deployment is key. Our refrigerated and freezer trailers can be set up quickly, often within hours, helping to minimize any disruption to your cold chain. Plus, our walk-in trailer units are designed for ease of use, typically running on standard 110-volt power. This means no complicated electrical hookups or expensive generators are usually required, making them incredibly practical for on-site use. And with walk-in access, loading, unloading, and organizing your pharmaceutical inventory becomes a breeze, which is a huge advantage when dealing with larger volumes. While our units are mobile, they are built to maintain precise temperatures, helping you adhere to the strict regulatory requirements for pharmaceutical products. You can easily integrate your own temperature monitoring and data logging systems for complete traceability and peace of mind.
We provide mobile refrigerated trailers that are perfect for pharmaceutical and chemical industries where raw ingredients and finished drugs need to be stored at precise temperatures. These units are built to withstand outside temperatures while keeping the inside just right. For more details on these adaptable options, explore our Mobile Cold Storage Solutions.
Frequently Asked Questions about Pharmaceutical Cold Storage
We know you might have some questions about keeping your vital medications safe and sound. Here are some of the most common ones we hear about pharmaceutical cold storage.
What happens if a pharmaceutical product is stored at the wrong temperature?
Think of pharmaceutical products like delicate plants – they need just the right environment to thrive! If a temperature-sensitive drug gets too hot or too cold, it can lose its power. This is called a "temperature excursion." When this happens, the medication might become less effective, or even completely useless. In some cases, it could even become harmful.
This isn't just bad for patient health; it also means big financial losses. Imagine a whole batch of life-saving medicine that can't be used! Improper storage can lead to wasted product, and that's a cost nobody wants. For patient safety, making sure drugs are stored correctly is absolutely critical.
How is temperature tracked in pharmaceutical cold storage units?
Keeping an eye on temperatures is super important in pharmaceutical cold storage. We use special tools called calibrated digital data loggers. Think of them as tireless little detectives that constantly check and record the temperature.
These systems are often hooked up to smart alarms. If the temperature starts to go outside the safe range, these alarms can send instant alerts to your phone or email. This means you know right away if there's an issue, so you can fix it fast. Having these detailed records creates a complete audit trail, which is a must for meeting all those important rules and regulations. It's all about making sure everything is exactly as it should be, every single second.
Can I get temporary cold storage for a pharmacy or clinic?
Absolutely, yes! Temporary cold storage is a lifeline for many healthcare facilities. At Icebox, we specialize in providing these flexible solutions. Our team of Mobile Refrigeration Specialists can deliver a refrigerated or freezer trailer right to your location.
These mobile units are perfect for all sorts of situations. Need extra space during flu vaccine season? Our mobile cooler trailers are ideal for handling that seasonal overflow. Doing renovations at your facility? A mobile refrigerated trailer can step in so your primary cold storage can be worked on without interruption. And, of course, they're fantastic as an emergency backup if your main equipment ever decides to take a day off.
We pride ourselves on offering reliable, 24-hour emergency service. Our walk-in trailer units are easy to set up and run on standard 110-volt power, making them super convenient for on-site use. Whether you're in Akron, Charlotte, Miami, Pittsburgh, or any of our other service areas along the East Coast, we're ready to bring the on-site cold storage solution you need, keeping your pharmaceuticals safe and sound.
Your Partner for Compliant and Reliable Cold Storage
When it comes to safeguarding pharmaceutical products, maintaining a precise cold chain isn't just important—it's absolutely critical for ensuring integrity and protecting public health. As the pharmaceutical world increasingly relies on temperature-sensitive medications, the old ways of doing things just won't cut it. Modern challenges call for flexible, quick, and highly responsive solutions that go beyond traditional, fixed storage options.
That's where we come in! Our Portable Cold Storage Units offer a truly practical, on-site solution for pharmacies, clinics, hospitals, and research facilities. Whether you're dealing with unexpected overflow, a sudden emergency, or planning a special project, these mobile units are designed to help you keep your valuable inventory safe. We understand that with pharmaceuticals, there's simply no room for error or delay.
That's why Icebox Mobile Refrigeration is dedicated to providing dependable, on-site mobile cold storage. Our service includes 24 Hour Cold Storage emergency support, because we know that sometimes, things just can't wait. Our easy-to-set-up, 110-volt walk-in trailer units are delivered directly to your location, ready to plug in and start protecting your temperature-sensitive products right away.
Imagine this: a sudden equipment breakdown, a busy flu season requiring extra vaccine storage, or a facility renovation that temporarily shuts down your main cold room. In these moments, our mobile refrigerated and freezer trailers seamlessly integrate into your operations, ensuring continuous temperature control without a hitch. We proudly serve a wide range of locations across the East Coast, including Akron, OH, Charlotte, NC, Fort Lauderdale, FL, Nashville, TN, and Pittsburgh, PA, among many others.
Don't let a temperature excursion compromise your critical pharmaceutical products or put patient safety at risk. Take a look at our Rentals today to find the perfect solution for your specific pharmaceutical cold storage needs. We’re here to help you keep your chill, no matter the challenge!
Choose the perfect rental option
Explore our wide range of trailer rentals and find the one that suits your needs
%20(2)-min.webp)
Discover New Blog Posts
Stay updated with our latest blog content.







